Mollusc survey add-on
Why molluscs?
Insects, molluscs and fungal pathogens can all have large impacts on plant communities but molluscs in particular have often been underestimated as drivers of plant community composition and functions. Furthermore, these groups can interact with each other (Allan & Crawley, 2011), and one mechanism driving such an interaction could be effects of insects and fungal pathogens on mollusc abundance. However, these effects have hardly ever been studies and we know almost nothing about how changes in insect or pathogen abundance might cascade to mollusc herbivores.
Molluscs are highly prevalent in many herbaceous communities and can have significant effects on plant composition. For instance, molluscs are often seedling predators that may reduce diversity by feeding on less competitive forb species (Overbeck et al., 2003). Insects and pathogens might therefore affect mollusc abundance by altering plant composition and promoting or reducing the favourite plants of molluscs. Insect herbivores and pathogens might also affect molluscs by altering plant chemical composition. There could even be direct interactions where fungi on leaves might attract or repel the molluscs (Ramsell & Paul, 1990). Further changes in biomass following insect herbivores or pathogen infection might alter microclimate or insects and pathogens could alter litter build-up, which might affect molluscs that feed on dead leaves (De Oliveira et al., 2010). Finally, molluscs might also affect insect and pathogen abundance through similar mechanisms, i.e., by altering plant composition, defence investment (Mann et al., 2021; Kästner et al., 2014), biomass and litter levels (Theenhaus & Scheu, 1996). But all of these are only hypotheses, and we still know very little about the molluscs.
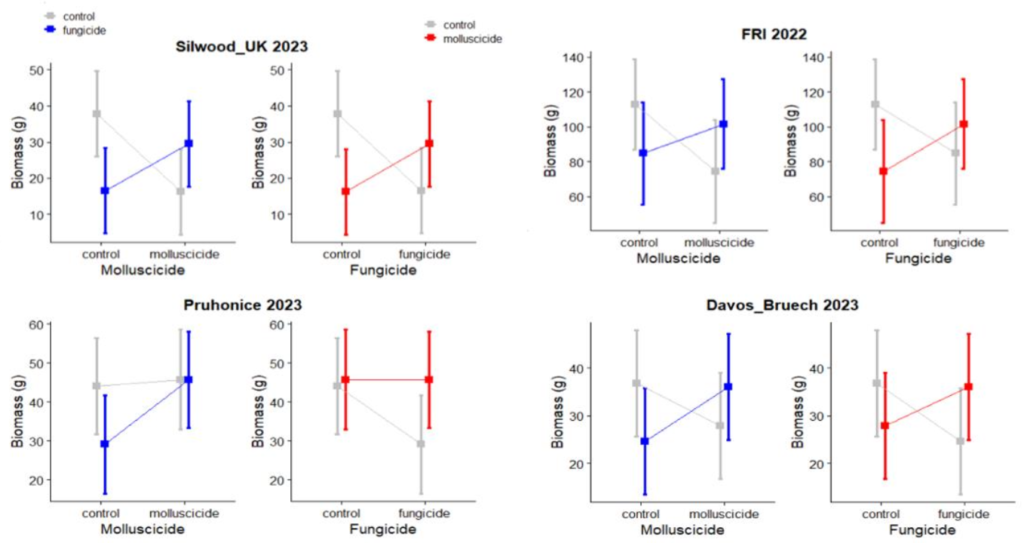
Very first results from analysing biomass from some of the BugNet experiments, show significant interaction effects of pathogens and molluscs on plant biomass. Strikingly, the same interaction was found in four separate experimental sites in different vegetation types (see figure below). I would therefore like to investigate this interaction further by exploring how mollusc abundance responds to changes in pathogens and insects in BugNet. I hope you share my excitement for exploring this and contributing to understanding the role of these neglected herbivores.
References
Allan, E., & Crawley, M. J. (2011). Contrasting effects of insect and molluscan herbivores on plant diversity in a long-term field experiment. Ecology Letters, 14(12), 1246–1253. https://doi.org/10.1111/j.1461-0248.2011.01694.x
De Oliveira, T., Hättenschwiler, S., & Tanya Handa, I. (2010). Snail and millipede complementarity in decomposing Mediterranean forest leaf litter mixtures. Functional Ecology, 24(4), 937–946. https://doi.org/10.1111/j.1365-2435.2010.01694.x
Kästner, J., Knorre, D. von, Himanshu, H., Erb, M., Baldwin, I. T., & Meldau, S. (2014). Salicylic Acid, a Plant Defense Hormone, Is Specifically Secreted by a Molluscan Herbivore. PLOS ONE, 9(1), e86500. https://doi.org/10.1371/journal.pone.0086500
Mann, L., Laplanche, D., Turlings, T. C. J., & Desurmont, G. A. (2021). A comparative study of plant volatiles induced by insect and gastropod herbivory. Scientific Reports, 11(1), Article 1. https://doi.org/10.1038/s41598-021-02801-2
Overbeck, G., Kiehl, K., & Abs, C. (2003). Seedling recruitment of Succisella inflexa in fen meadows: Importance of seed and microsite availability. Applied Vegetation Science, 6(1), 97–104. https://doi.org/10.1111/j.1654-109X.2003.tb00568.x
Ramsell, J., & Paul, N. D. (1990). Preferential Grazing by Molluscs of Plants Infected by Rust Fungi. Oikos, 58(2), 145–150. https://doi.org/10.2307/3545421
Theenhaus, A., & Scheu, S. (1996). The influence of slug (Arion rufus) mucus and cast material addition on microbial biomass, respiration, and nutrient cycling in beech leaf litter. Biology and Fertility of Soils, 23(1), 80–85. https://doi.org/10.1007/BF00335822
Protocol
We propose to collect molluscs with shelter traps. They should provide protection for all kinds of molluscs without attracting those from surrounding plots. In addition, we ask you to measure vegetation height to get an idea of microclimate conditions. After molluscs’ collection please send sampling tubes back to us. In the following we will describe the sampling and data procedure in detail.
Required material
Shelter trap
Material provided by us:
- 48 35 cm x 35 cm cardboards (2 per plot)
- 48 Falcon tubes
- 48 zipper plastic bags
To be supplied by collaborators:
- Nails to stabilize the board (or other sharp material)
- Ethanol
Building shelter traps
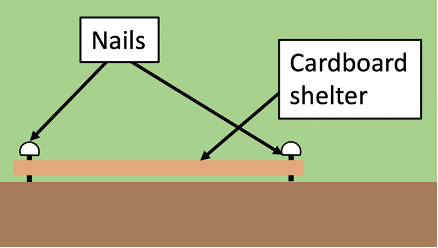
SHELTER: a cardboard with tape at the side (to prevent small molluscs from hiding in the cavities of the corrugated cardboard) should be placed directly on the ground. Add nails in the four corners of the traps.
Placing the traps in the field
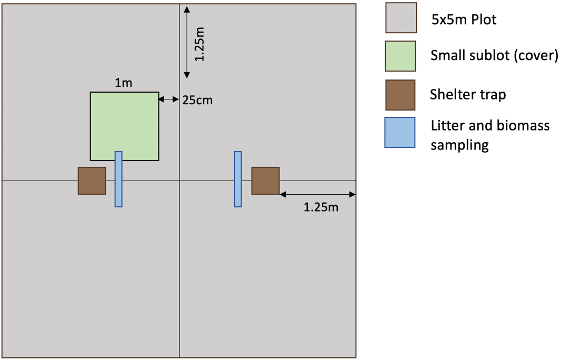
The traps have to be placed in the centre of the plot, between the subplots, to avoid edge effects. If you are performing a site-specific treatment in one subplot that involves chemicals (e.g. a mycorrhizal killer) move the traps slightly away from that subplot. Each trap has to be installed twice within a plot: 48 shelter traps in total (Fig. 3).
Mollusc sampling
Timing
The best time to install the traps would be in spring or autumn where the weather conditions are mild or wet, therefore more adequate for the molluscs. Ideally install the traps shortly before, during, or after a rain event.
Shelter trap
Collect the molluscs from the traps first time one week after setting up the traps, and a second time two weeks after setting up the traps. The molluscs should be still alive, but they have to be put in ethanol tubes. You can pool the molluscs across the two shelter traps per plot. Please use one tube per plot for collection 1 and another tube per plot for collection 2.
Labelling and sending the sampling tubes
Label each tube carefully at the outside and inside. At the outside use a sticky label (laser printers are alcohol proof) or a waterproof (ideally ethanol proof) marker. At the inside either add a label printed in alcohol proof ink, or written in lead pencil as pen ink and some printer inks will rub off if any ethanol leaks from the tubes. Write down the following information:
Sending molluscs to Bern
After collection, send molluscs back to Bern. Make sure tubes are well closed, and place all tubes in an airtight container (e.g. a ziplock bag). Send tubes to this address (please inform us in advance):
Institute of Plant Sciences
Eric Allan / Eva Demont Alternbergrain 21
3013 Bern
Switzerland
If it is not possible to send mollusc samples to Switzerland, but you would still like to participate in the mollusc add-ons or you have questions, please get in touch with us. We can explain you the measurement procedure in detail.
Add-on measures
In addition to the mollusc trapping, it would be great to take a couple of further measures, however, there are not necessary and you can still participate in the add-on without them.
Vegetation height: as a proxy of local microclimate, we would like collaborators to measure the maximum height of the vegetation (without stretching or lifting up stems) around the traps. Measure the vegetation height 20 cm above and below each trap along the middle lines with a ruler (8 measures in total per plot).
Humidity rate: another variable of microclimate is the humidity level around the traps. To do so, after the last mollusc harvest, the wet cardboards can be weighed, then dried in an oven, and weighed again. Provided zipper plastic bags can be used to transport the cardboards from the field to the lab to maintain humidity.
Vegetation cover: take a picture of each plot to estimate the cover of the vegetation. Simple pictures took with a phone at 1/1.5m above the soil are fine.
Herbivore damages: it would be nice to have damage data from the same year in which we survey molluscs. If you have the time it would therefore be nice to measure herbivore and pathogen damage according to our leaf damage protocol.
Litter biomass at mollusc collection: Take two 10 cm x 50 cm biomass samples (or one 20 x 50 cm sample) next to each trap (Figure 3) during the period in which the molluscs are being trapped, pool them and sort the biomass into live and dead biomass. Dry and weigh the dead and life parts separately per plot, and add the live and dead mass into the mollusc add-on datasheet (mass_sampleC).
Litter biomass at peak biomass: Also sort the main biomass of the plots (collected at peak biomass) into live and dead biomass and weight it separately. Add the live and dead mass data separately to the mollusc add-on datasheet, and the total biomass data as always to the experimental data sheet.

